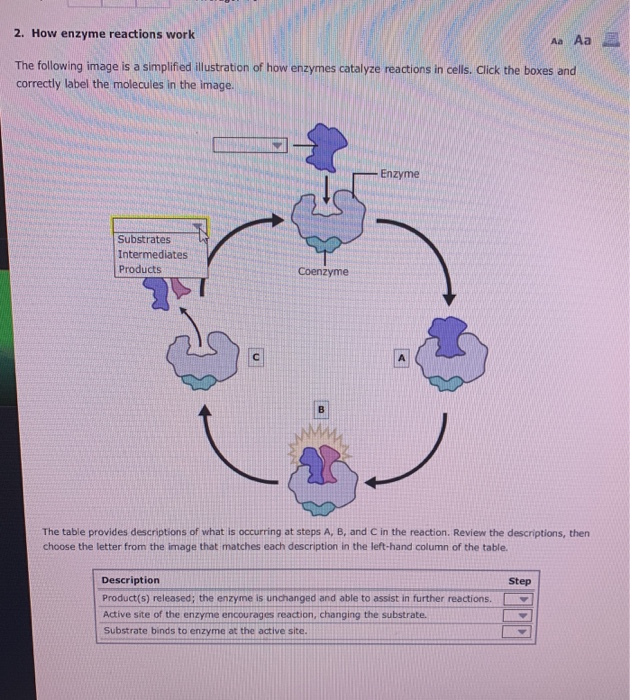
Graphically, the results of these experiments are shown above. At low concentrations of substrate, the inhibitor competes for the enzyme effectively, but at high concentrations of substrate, the inhibitor will have a much reduced effect, since the substrate outcompetes it, due to its higher concentration (remember that the inhibitor is at fixed concentration). Next, a second set of reactions is performed in the same manner as before, except that a fixed amount of the methotrexate inhibitor is added to each tube. 
reactions without inhibitor (20 or so tubes, with buffer and constant amounts of enzyme, varying amounts of substrate, equal reaction times). Conversely, the more folate there is, the less of an effect methotrexate has on the enzyme because folate outcompetes it.įigure 5.4.1: Competitive Inhibition Figure 5.4.2: Line-Weaver Burk Plot of competitive inhibition The more methotrexate there is, the more effectively it competes with folate for the enzyme’s active site. As a result, methotrexate ‘competes’ with folate for binding to the enzyme. Notably, the binding site on DHFR for methotrexate is the active site, the same place that folate would normally bind. When the drug methotrexate is present, some of the enzyme binds to it instead of to folate and during the time methotrexate is bound, the enzyme is inactive and unable to bind folate. 
This enzyme normally catalyzes the reduction of folate, an important reaction in the metabolism of nucleotides. An example is methotrexate, which resembles the folate substrate of the enzyme dihydrofolate reductase (DHFR). Molecules that are competitive inhibitors of enzymes resemble one of the normal substrates of an enzyme. Probably the easiest type of enzyme inhibition to understand is competitive inhibition and it is the one most commonly exploited pharmaceutically.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
